When it comes to identifying proteins in research or developing therapeutics for clinical use, antibodies have become uniquely placed as the most suitable option. With the availability of multiple options, how do you decide whether a monoclonal or polyclonal antibody is right for your experiment?
What is an antibody?
Antibodies are proteins secreted by B cells in the immune system to recognize, bind, and neutralize antigens that are unique to organisms such as viruses and bacteria. Antibodies are likely the lifeline of scientific, clinical research and diagnosis due to their role in antigen detection and identification.
You can easily get the custom rabbit monoclonal antibody discovery service via Boster Bio.
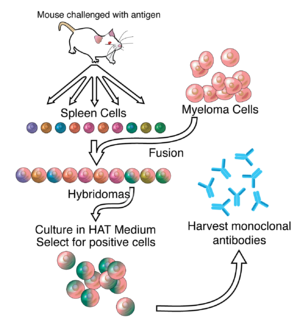
Image Source: Google
These antigens are usually proteins, but can also be nucleotides, small molecules, or macromolecules. The ability of an antibody to bind to an antigen effectively depends on the antigen surface-binding sequence, known as an epitope, and how well the epitope matches the antibody-binding surface sequence.
Antibodies are structured as Y-shaped molecules, with the tail end responsible for effector function and allowing the antibody to interact with other cells of the immune system. Each Y arm has a variable region that is specific for epitope recognition on antigens. Read more about our brief history of antibodies here.
What is the difference between monoclonal and polyclonal antibodies?
When animals are injected with an immunogen (antigen), the cells of the primary immune response secrete various antibodies that recognize and bind to multiple epitopes of the antigen. Since the secreted antibodies are produced by several plasma cells, they are called polyclonal antibodies.
Monoclonal antibodies, on the other hand, must be produced by the same B cells that are clones of the parent cell. This means that monoclonal antibodies can only recognize and bind to the epitope of the same antigen, thereby reducing the risk of cross-reactivity. Polyclonal antibodies are usually made in vivo and monoclonal antibodies are made ex vivo, so they are easier to standardize.